What Is The Triple Point Of Water?
The triple point of water is the exact temperature and pressure where ice, liquid water, and water vapor coexist in equilibrium. It occurs at 0.01°C (273.16 K) and 611.657 pascals (0.006 atm). This unique condition enables key processes in commercial freeze drying and food processing.
Quick Overview
At the triple point, water phases balance perfectly, allowing direct ice‑to‑vapor sublimation without melting. Freeze dryers operate just below this point for efficient dehydration. Microwave drying avoids the triple point altogether, using vacuum and volumetric heat for faster, energy‑saving alternatives in food tech.
What Defines The Triple Point Of Water?
The triple point marks where solid, liquid, and gas phases of water meet stably. Only at 0.01°C and 611.657 Pa do all three exist together. Any shift in temperature or pressure tips the balance to one dominant phase.
Thermodynamic equilibrium holds there. Molecules shift freely between ice, water, and vapor without net change.
Why Is The Triple Point Important In Everyday Science?
The triple point calibrates temperature scales. Kelvin defines 273.16 K as the triple point. International standards use a water triple point cell for precision measurement.
It also sets benchmarks for pressure gauges and phase studies.
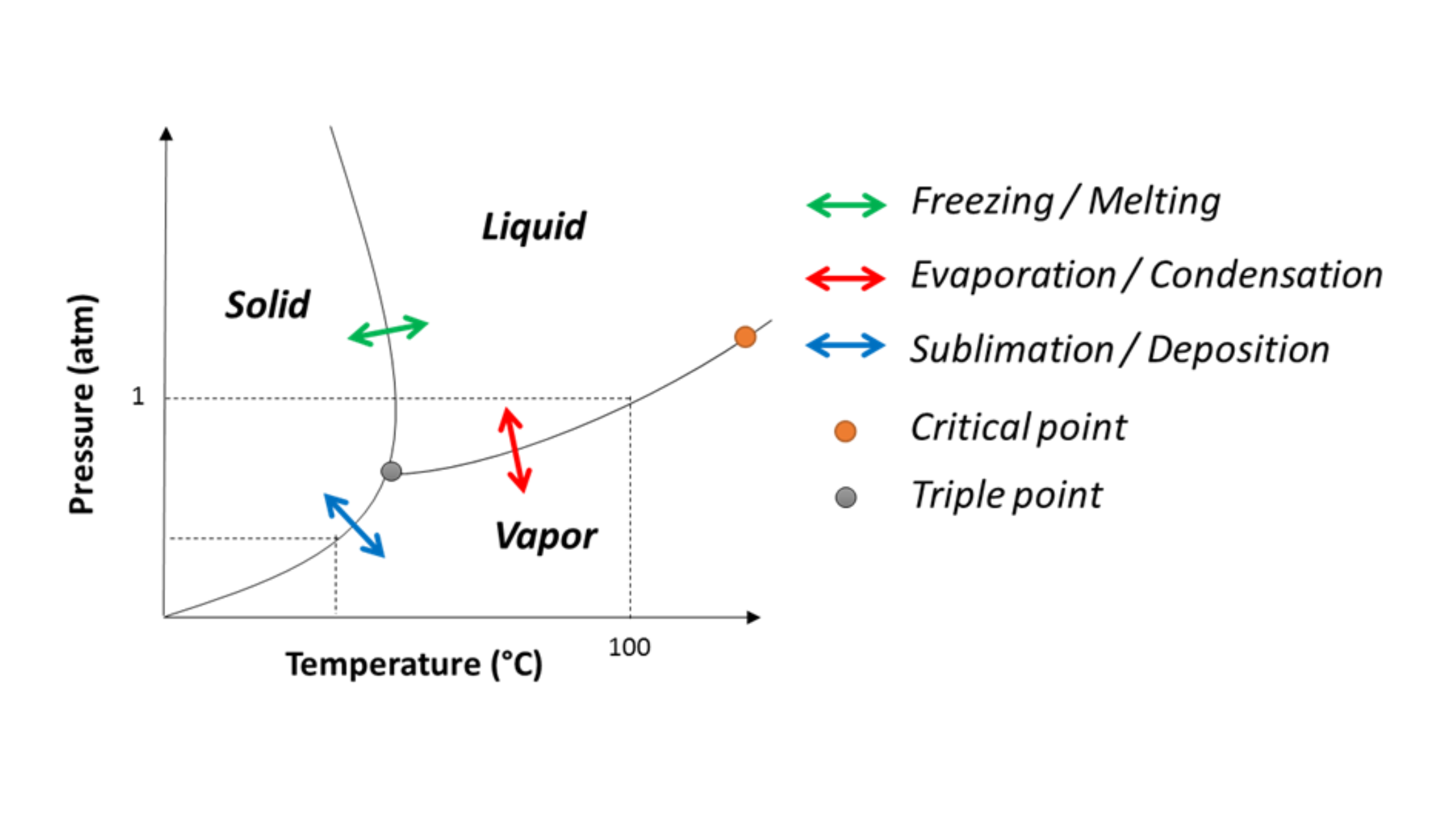
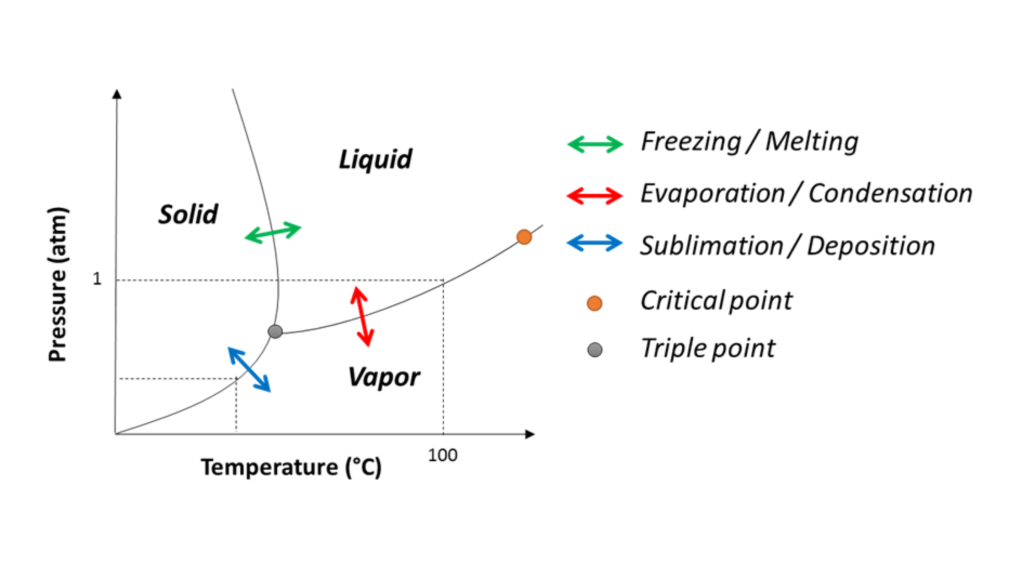
How Does The Triple Point Appear On A Water Phase Diagram?
Water phase diagrams plot pressure versus temperature. Three lines meet at the triple point. One shows melting/freezing, one boiling/condensation, one sublimation/deposition.
Below the triple point pressure, ice sublimes directly to vapor. Above, liquid forms first.
What Role Does The Triple Point Play In Freeze Drying?
Freeze drying relies on conditions near the triple point. Product freezes solid, then vacuum lowers pressure below 611 Pa. Heat drives sublimation from ice to vapor, bypassing liquid.
Commercial freeze drying chambers control pressure around 100–300 Pa and temperatures just above ‑40°C. That maximizes sublimation rate without melt‑back.
Why Must Freeze Dryers Stay Below The Triple Point Pressure?
Pressure below triple point prevents liquid formation. At triple point or above, ice melts before subliming. Collapse ruins structure.
Dryers pull 50–200 Pa vacuum. Condensers trap vapor as ice, maintaining low pressure.
How Does Temperature Control Work Near The Triple Point?
Shelf temperatures rise gradually during primary drying. Start at ‑40°C, increase to ‑20°C or higher as ice sublimes. Product temperature stays below collapse point.
Sensors monitor chamber pressure and shelf heat. Recipes optimize for each product.
What Happens At Exactly The Triple Point In A Dryer?
At precise triple point, all phases coexist briefly. Practically, dryers avoid it to favor sublimation. Small liquid pools could form and refreeze unevenly.
Equilibrium slows net drying. Operations stay below for efficiency.
Why Does The Triple Point Make Water Unique?
Water’s triple point lies above 0°C freezing at standard pressure. Hydrogen bonding expands ice, altering phase behavior. Most substances have triple points below freezing.
This anomaly aids freeze drying. Sublimation occurs readily under vacuum.
How Does Triple Point Relate To Food Processing Challenges?
High‑sugar or fat foods have depressed triple points. Eutectic mixtures freeze lower. Dryers must map each product’s phase diagram.
Custom recipes prevent collapse. Testing determines safe temperatures and pressures.

What Common Mistakes Occur Near The Triple Point In Drying?
Operators overshoot shelf heat, causing melt‑back. Vacuum leaks raise pressure above safe levels. Ignoring product eutectic leads to liquid pockets.
Monitor with Pirani gauges and product thermocouples. Ramp heat slowly.
How Do Modern Freeze Dryers Optimize Triple Point Operations?
Lyostats and manometric temperature measure sublimation directly. Software adjusts shelves automatically. PAT tools predict endpoint.
Advanced controls cut cycles 20–30 percent.
Why Consider Microwave Drying Equipment Over Triple Point Reliance?
Microwave drying uses vacuum but skips triple point freezing. Volumetric heat evaporates water rapidly at low temperatures. Cycles run 30–90 minutes versus days.
No condensers or deep freezing needed. Energy drops.
How Does REV™ Microwave Drying Avoid Triple Point Limits?
REV™ operates under vacuum with microwave energy. Water boils at low temperature without freezing. Uniform heating prevents wet cores.
Food processors gain throughput for product innovation.
What Advantages Does Microwave Offer Food Dehydration?
Microwave drying suits high‑volume snacks and ingredients. Texture control from crispy to chewy. Lower capex than large freeze dryers.
Hybrid lines combine both for flexibility.
Triple Point Checklist For Freeze Drying Success
Follow these steps.
- Map product eutectic point.
- Freeze below triple point temperature.
- Pull vacuum to 100–300 Pa.
- Ramp shelf heat gradually.
- Monitor product temperature.
- Confirm dry state with pressure rise test.
- Desorb bound water in secondary phase.
FAQ
What is the triple point of water?
The triple point of water is 0.01°C (273.16 K) and 611.657 Pa where ice, liquid, and vapor coexist in equilibrium.
Why is the triple point important for freeze drying?
It enables sublimation below this pressure. Dryers operate near it to turn ice directly to vapor without liquid.
What temperature and pressure is water’s triple point?
0.01°C and 611.657 pascals (0.006 atm). This defines the Kelvin scale.
Can liquid water exist below the triple point?
No. Below triple point pressure, ice sublimes to vapor without melting.
How does the triple point appear on phase diagrams?
As the intersection of solid‑liquid, liquid‑gas, and solid‑gas lines.
Why do foods have different triple points?
Solutes depress the point. Eutectic mixtures freeze lower than pure water.
What happens if pressure exceeds triple point in drying?
Ice melts to liquid, causing collapse and poor structure.
Do microwave dryers use the triple point?
No. They evaporate water under vacuum without freezing, using heat directly.
How precise must freeze dryers control near triple point?
Within 10–50 Pa pressure and 1°C temperature for optimal sublimation.
What defines thermodynamic equilibrium at triple point?
No net phase change. Molecules shift between phases without overall loss or gain.
Sources and further reading
Triple Point of Water Definition
Phase Diagram of Water
https://byjus.com/chemistry/phase-diagram-of-water
Science Notes Triple Point
Millrock Tech Freeze Drying Explained
https://www.millrocktech.com/lyosight/lyobrary/what-is-freeze-drying
Purdue Chemistry Phase Diagrams
https://www.chem.purdue.edu/gchelp/liquids/pliqgas.html
Fluke Calibration Triple Point
https://www.fluke.com/en-ca/learn/blog/calibration/what-is-triple-point-of-water
EnWave Exploring Alternatives to Freeze Drying
EnWave Freeze Drying Vs Microwave Drying
Food Processing Drying Technologies